Electrical Charge, Current,
Potential difference and Power
Electric charge is a property possessed by protons and electrons which creates an electric field, which is a region of space in which forces are exerted on other bodies with charge. Like charges repel each other, while unlike charges attract each other. Protons have a positive charge while electrons have a negative charge of equal size. Electric charge is given the symbol Q and has the SI units of the coulomb, C. The charge on one electron is 1.6 × 10-19 C. This value is given the special symbol é, where the accent indicates it is a negative charge.
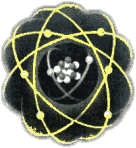 Atoms are made up of a positively charged nucleus (containing protons and neutrons) about which the negatively charged electrons orbit. It is the interaction of electric fields that hold the electrons to the nucleus. There are equal numbers of protons and electrons which means the atom is overall neutrally charged.
Atoms are made up of a positively charged nucleus (containing protons and neutrons) about which the negatively charged electrons orbit. It is the interaction of electric fields that hold the electrons to the nucleus. There are equal numbers of protons and electrons which means the atom is overall neutrally charged.
If you give an electron more energy then it is able to leave the nucleus, in much the same way a rocket can leave the earth’s gravity field. This can be caused by collisions with atoms or particles, the effect of radiation, an electric field or chemical action. This process is known as ionisation, and creates positive and negative ions, according to their electric field.
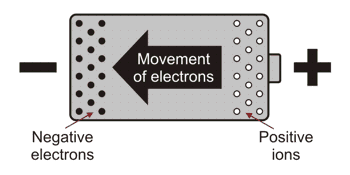
In a cell, chemical energy is used to separate electrons from atoms. One terminal becomes deficient in electrons and therefore becomes positive. While the other becomes negative as it acquires an excess of electrons. You could picture an unconnected cell as having its negative terminal crowded with electrons. As like charges repel each other, these electrons are ‘keen’ to escape from the terminal. The positive terminal has a number of positive ions which are desperately seeking electrons to ‘neutralise’ them.
Metals are very good conductors of electricity. This is due to their internal structure. The atoms are arranged in lattice, which share the outermost electrons. These electrons are free to move between the different positively charged ions (nuclei). You can think of this as a ‘sea of electrons’ which act as glue to hold the ions together. At all temperatures above absolute freezing the positive ions are vibrating in their place, with the ‘free’ electrons move rapidly in random directions.

When a metal wire is placed between the terminals of a cell the free electrons in the metal are repelled by the concentration of electrons at the negative terminal. While at the positive terminal the free electrons in the conductor are ‘sucked’ into the positive ions. This then leaves a deficiency of electrons in the conductors which attracts more electrons towards it. At the other end the ‘crowd’ of electrons ‘spill’ out of the cell, pushing the free electrons in front of them. In this way an electrical signal can travel much faster than the individual electrons. In fact the signal travels very nearly at the speed of light.
In a short period of time, the distribution of electrons evens out into a steady flow all around the circuit. The rate of flow of electrically charged particles is called electrical current. Electrical current is given the symbol I and has the SI units of the Ampere, A.
"Rate of" means how much per bit of time, so the equation to calculate current is as follows:
|
|
where |
I |
= |
Current in Amperes, A |
| Q |
= |
Charge in Coulombs, C |
| t |
= |
Time in Seconds, s |
|
From this equation we can see that a Coulomb is the amount of electrical charge that passes a given point in a circuit when a current of 1A flows for 1s.
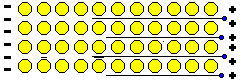
The free electrons are accelerated by the electric field, gaining kinetic energy. When an electron collides with an ion, some of its kinetic energy is transferred to the ion as vibrational energy, resulting in an increase in the temperature of the metal. This heat energy is then lost to the surroundings. Energy has left the circuit.
The potential difference (p.d.) between two points in a circuit is defined as the amount of electrical energy changed to other forms of energy, per coulomb of charge flowing between them. Potential difference is given the symbol V and has the SI units of volts, V.
One volt is the p.d. between two points in a circuit in which 1 joule of energy is converted to other forms when 1 coulomb of charge passes between them. So the equation to calculate p.d. is as follows:
|
|
where |
V |
= |
Potential difference in Volts, V |
| E |
= |
Energy in Joules, J |
| Q |
= |
Charge in Coulombs, C |
|
Power is the rate of delivery of energy so:
|
|
where |
P |
= |
Powers in Watts, W |
| E |
= |
Energy in Joules, J |
| t |
= |
Time in Seconds, s |
|
These equations can be combined as follows:
Metals are not the only things to conduct electricity. It is possible for fluids to become ionised and therefore able to carry current eg a neon lamp. Given enough energy it is possible for electrons to leave metal surfaces. This needs a very large potential difference. We can then create an electron beam which itself is a current. The current is equal to the number of particles arriving per second multiplied by the charge on one particle.
|
I = NQ |
where |
I |
= |
Current in Amperes, A |
| N |
= |
Number of ions arriving per second |
| Q |
= |
Charge of one ion in Coulombs, C |
|
The energy supplied per second (power of the beam) is the energy of one ion multiplied by the number of ions arriving per second:
|
P = NE = NQV |
where |
P |
= |
Powers in Watts, W |
| N |
= |
Number of ions arriving per second |
| E |
= |
Energy of one ion in Joules, J |
| Q |
= |
Charge of one ion in Coulombs, C |
| V |
= |
Potential difference in Volts, V |
|
 Atoms are made up of a positively charged nucleus (containing protons and neutrons) about which the negatively charged electrons orbit. It is the interaction of electric fields that hold the electrons to the nucleus. There are equal numbers of protons and electrons which means the atom is overall neutrally charged.
Atoms are made up of a positively charged nucleus (containing protons and neutrons) about which the negatively charged electrons orbit. It is the interaction of electric fields that hold the electrons to the nucleus. There are equal numbers of protons and electrons which means the atom is overall neutrally charged.


